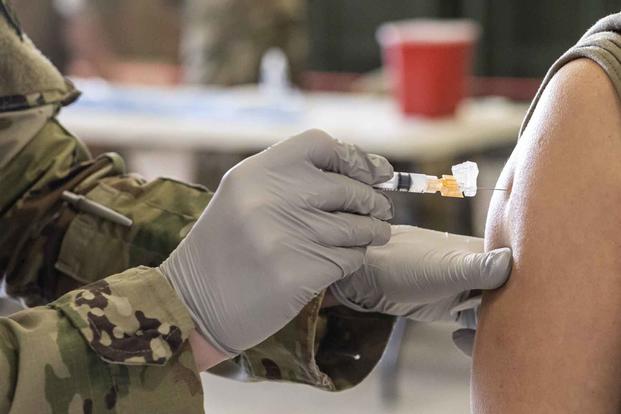
The Defense Department has waived a requirement that Tricare Prime patients have a referral from their primary care doctors to get the COVID-19 vaccine outside the military health system.
The waiver, to be published in the Federal Register on Tuesday, allows Prime enrollees to receive the vaccine at any non-military facility, including non-network pharmacies or providers, without incurring any fees.
Read Next: Air Force Instructor Pilot, Japanese Student Killed in Alabama T-38 Crash
While the vaccine is free to all Americans, some providers charge patients an administrative fee, much like a charge for a doctor's visit. The charges are reimbursable for those with medical insurance, but patients must file a claim with their insurers to recoup the payments.
The waiver will insure that Tricare Prime beneficiaries don't incur any charges if they get their vaccine from a location other than their military health provider. Ordinarily Tricare beneficiaries must get a referral for outside care through their primary care physician or pay point-of-service charges.
Officials have encouraged Tricare beneficiaries to seek the vaccine through their local or state public health departments or at hospitals or pharmacies offering it, including Walgreens, which is in Tricare's retail pharmacy network, and the non-network CVS pharmacies.
The waiver will remain in effect throughout the national emergency declared for the coronavirus pandemic.
The waiver does not apply to any other vaccines at non-network pharmacies.
As of Monday, More than 75 million COVID-19 vaccines have been delivered to states, localities and federal entities nationwide, and 63 million doses have been administered, including nearly 19 million second doses.
Across the Defense Department, 854,728 doses of the 1.18 million received by the department have been administered. Roughly 233,600 military-connected people have received their second dose -- namely active-duty personnel and civilian employees who work in health care, emergency services or billets of national security importance, and Tricare beneficiaries ages 75 and older.
Some locations across DoD have started administering vaccines to those 65 and older, and those under 65 with underlying health conditions.
Roughly 250,000 of the nation's 28 million COVID-19 cases have been among active-duty service members, civilian DoD employees, family members or contractors. Among the military-affiliated cases, 288 have resulted in death.
A Centers for Disease Control and Prevention report released last week found that the COVID-19 vaccines manufactured by Pfizer-BioNTech and Moderna had relatively low rates of adverse events. Among the 13.7 million vaccines administered between Dec. 14 and Jan. 13, there were 6,994 adverse event reports filed with the agency, nearly 91% of which were categorized as "non-serious."
The most common non-serious adverse events included headache, fatigue and dizziness, reported most frequently after the second dose.
The report also noted that, of the adverse events among those not living in nursing homes or other long-term care facilities, nearly 81% were reported from the Pfizer-BioNTech vaccine, while 19.2% were reported from the Moderna vaccine.
There have been 113 deaths reported to the Vaccine Adverse Event Reporting System, roughly 65% of which occurred in long-term care facilities. However, the CDC said that information from death certificates, medical records and other resources did not link the vaccines as the cause of death.
The rate of anaphylaxis -- a severe allergic reaction that remains a concern related to the new vaccines -- was 4.5 cases per million doses administered, or 62 patients. That includes 46 with the Pfizer vaccine and 16 from the Moderna vaccine.
-- Patricia Kime can be reached at Patricia Kime@Monster.com. Follow her on Twitter @patriciakime.
Related: Tricare Users Frustrated By Lack of Information on COVID-19 Vaccine Availability